Tetsuya Tajima, United States has been granted the TTS Scientific Congress Award

Dr. Tetsuya Tajima completed his 6-year surgery residency and joined the Division of Hepato-Biliary-Pancreatic Surgery and Transplantation at Kyoto University, Japan as a surgical instructor in 2016. The division started a liver transplantation program in 1990, and to date, it has performed more than 1,900 living-donor liver transplants, more than any other institution in Japan. Dr. Tajima has been involved in both basic and clinical research in liver transplantation. In 2021, Dr. Tajima obtained his Ph.D. and in 2022 an M.P.H. at Kyoto University, respectively.
Dr. Tajima joined Krams and Martinez Labs (Transplant Immunology Labs), at Stanford, as a Postdoctoral Scholar in April 2022 and was awarded an internal Transplant and Tissue Engineering Center of Excellence Fellowship (stipend support) for his studies examining refractory rejection after liver transplant in an experimental model. In addition, Dr. Tajima is working on the DoD-funded project where he is using high-dimensional mass cytometry (CyTOF) to discern the functional significance of specific myeloid and T cell populations during rejection in a mouse model of vascularized composite allotransplantation (VCA). Besides, Dr. Tajima is conducting clinical research on Epstein-Barr virus (EBV)-associated post-transplant lymphoproliferative disorders (PTLD) and the impact of induction therapy on rejection in pediatric transplantation using data from a prospective multicenter study in the United States, which was supported by a grant from the National Institutes of Health. Dr. Tajima was the recipient of the Japan Society for Transplantation Award 2022 for his clinical research achievements.
Epstein-Barr virus-associated post-transplant lymphoproliferative disorders in pediatric transplantation: A prospective multicenter study in the United States
Tetsuya Tajima1, Daniel Bernstein2, Scott D. Boyd3, Dita Gratzinger3, Grant Lum1, Kazunari Sasaki1, Brent Tan3, Clare J. Twist4, Kenneth Weinberg2, Mark A. Robien5, Merideth Brown5, Brian Armstrong6, Dev Desai7, George Mazariegos8, Clifford Chin9, Thomas M. Fishbein10, Akin Tekin11, Robert S. Venick12, Olivia M. Martinez1, Sheri M. Krams1, Carlos O. Esquivel1.
1Department of Surgery, Division of Abdominal Transplantation, Stanford University School of Medicine, Stanford, CA, United States; 2Department of Pediatrics, Stanford University School of Medicine, Stanford, CA, United States; 3Department of Pathology, Stanford University School of Medicine, Stanford, CA, United States; 4Department of Pediatric Oncology, Rosewell Park, Buffalo, NY, United States; 5National Institute of Allergy and Infectious Diseases, Rockville, MD, United States; 6Rho, Durham, NC, United States; 7Division of Surgical Transplantation, UT Southwestern Medical Center, Dallas, TX, United States; 8Department of Pediatrics, UPMC Children’s Hospital, Pittsburgh, PA, United States; 9Department of Pediatrics and Cincinnati Children’s Hospital, University of Cincinnati, Cincinnati, OH, United States; 10Departments of Surgery and Pediatrics, MedStar Georgetown University Hospital, Washington, DC, United States; 11Department of Surgery, University of Miami Miller School of Medicine, Miami, FL, United States; 12Department of Pediatric Gastroenterology, David Geffen School of Medicine, University of California Los Angeles, Los Angeles, CA, United States
Clinical Trials in Organ Transplantation in Children (CTOTC-06).
Introduction: Epstein-Barr virus (EBV)-associated post-transplant lymphoproliferative disorders (PTLD) is the most common malignancy in children after transplant; however, early detection is still challenging, which may result in a worse prognosis.
Methods: This prospective, multicenter, study enrolled 944 children (≤ 21 years of age). Of these, 872 received liver, heart, kidney, intestinal, or multivisceral transplants in 7 US centers between 2014, and 2019 (NCT02182986). Participants were followed for a minimum of 12 months and up to four years based on the time of enrollment. In total, 34 pediatric EBV+ PTLD (3.9%) were identified by biopsy. Sex, age, race, ethnicity, transplanted organ, EBV viral load, pre-transplant EBV serology of donor and recipient, immunosuppression, response to chemotherapy and rituximab, and histopathological diagnosis were analyzed.
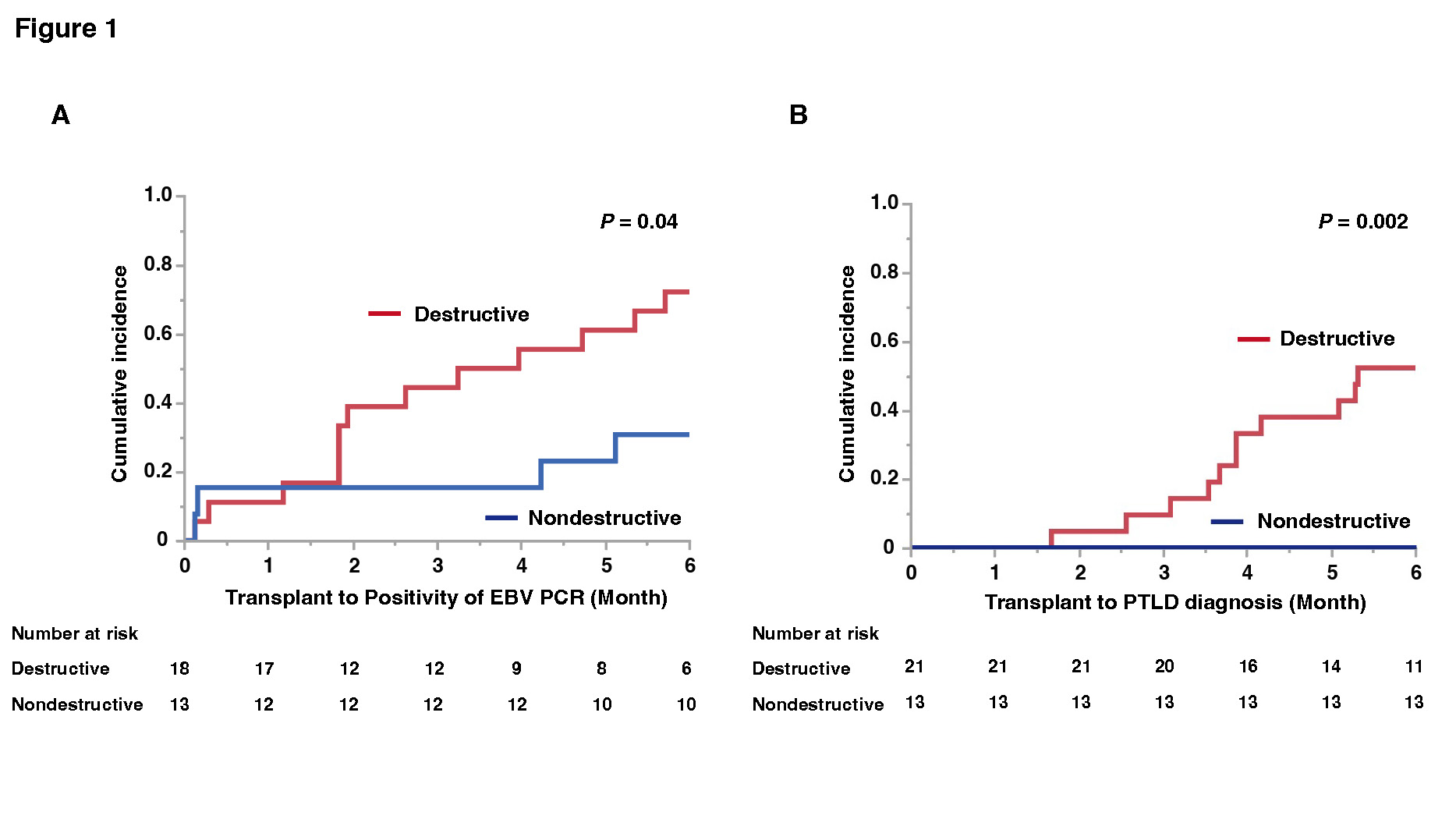
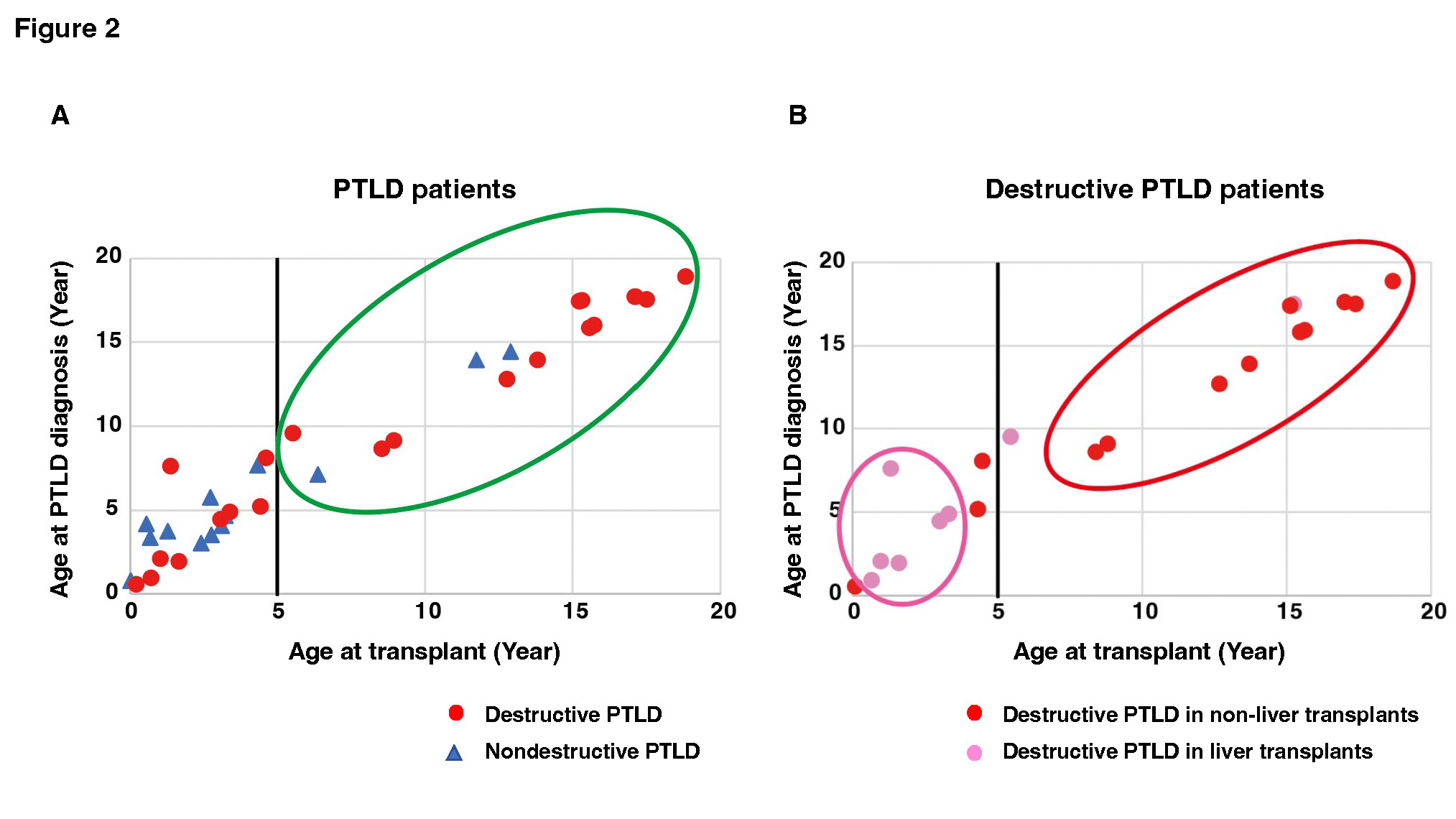
Results: Eighteen male and 16 female patients developed EBV+ PTLD, and the median ages at transplant and PTLD diagnosis were 4.3 years (IQR: 1.4–13.0) and 4.4 years (IQR: 2.6–12.8), respectively. The number of transplanted organs was 12 livers, 8 hearts, 7 kidneys, and 7 intestines including multiple organs. The 1-year, 2-year, and 3-year survival rates of patients after PTLD diagnosis were 90%, 83%, and 83%, respectively. The entire cohort was divided into two cohorts: 21 patients with destructive PTLD (monomorphic and polymorphic) and 13 patients with nondestructive PTLD (early lesion). Ages at transplant and PTLD diagnosis were significantly higher in destructive than in nondestructive PTLD patients (P = 0.03 and P = 0.04, respectively). Destructive PTLD patients had more extranodal lesions (P < 0.001) and were more frequently treated with chemotherapy or rituximab than nondestructive PTLD patients (P = 0.01). Destructive PTLD patients had detectable EBV by PCR more frequently than nondestructive PTLD patients within six months after transplant (P = 0.04, Figure 1-A), and EBV viral load at PTLD diagnosis was significantly higher in destructive than in nondestructive PTLD patients (P = 0.04). Pediatric recipients developed significantly more destructive PTLD than nondestructive PTLD within six months (P = 0.002, Figure 1-B). Patients ≥ 5 years of age at transplant had significantly more destructive than nondestructive PTLD (P = 0.047, Figure 2-A). Moreover, among patients with destructive PTLD, there were significantly more liver transplant recipients at age ≤ 5 years at transplant and non-transplant recipients at age ≥ 5 years at transplant (P = 0.02, Figure 2-B).
Conclusion: Destructive PTLD had significantly earlier onset and higher EBV viral load within six months post-transplant than nondestructive PTLD. Close follow-up of EBV within six months after transplant, especially in children ≤ 5 years at liver transplants or ≥ 5 years at non-liver transplants, may facilitate early detection of destructive PTLD.
NIH 1UO1AI104342. Transplant and Tissue Engineering Center of Excellence.