
Belatacept outcomes in pediatric kidney transplantation in France
Charlotte Duneton1, Elodie Cheyssac1, Houaida Dahdouh1, Véronique Baudouin1, Olivier Dunand2, Anne Laure Sellier-Leclerc3, Julien Hogan1.
1Pediatric Nephrology Department, , Robert Debré University Hospital AP-HP, Paris, France; 2Pediatric Nephrology Department, Félix Guyon University Hospital, La Réunion, France; 3Pediatric Nephrology Department, Mother-Child University Hospital, Lyon, France
Purpose: Belatacept (CTLA-Ig) is a selective co-stimulation blocker that is associated with reduced dnDSA, improved renal function, and prolonged allograft survival in adult transplant recipients compared to calcineurin inhibitor (CNI) based regimens. Its use in older children and young adults is limited. We report outcomes for 18 pediatric patients converted to belatacept.
Methods: 18 patients converted to belatacept therapy between 05/2018 and 01/2022 were included in 3 pediatric nephrology departments in France (Robert Debré Paris, Lyon and La Réunion). Patients received an induction with basiliximab (n=12) or antithymocyte globulin (n=6). Maintenance immunosuppression included CNI, antimetabolite and steroids. Patients’ viral status (EBV, CMV) were monitored monthly and allograft biopsy was performed prior to conversion and 6 months after conversion. The first 5 belatacept injections were administered at 5mg/kg/dose every 2 weeks, then monthly. CNI doses were decreased by 25% at each infusion and stopped after 2 months. Antimetabolite doses were also increased at CNI withdrawal. 6/18 patients were steroid-free at the time of conversion.
Results: The median age at conversion was 13,5 years (range 11,4-15,2) and 3,7 years (IQR 1,9 – 5,9) post-transplant. Conversion indication was based on medical need for long term CNI avoidance (side-effect or histological evidence of CNI toxicity, n=11) or to improve adherence (e.g. monthly IV-treatment, n=7). CNI was withdrawn in 17/18 patients by a median of 42 days (IQR 42-75) after starting belatacept. GFR was stable or improved over a median follow-up time of 12 months (IQR 8-17,2).
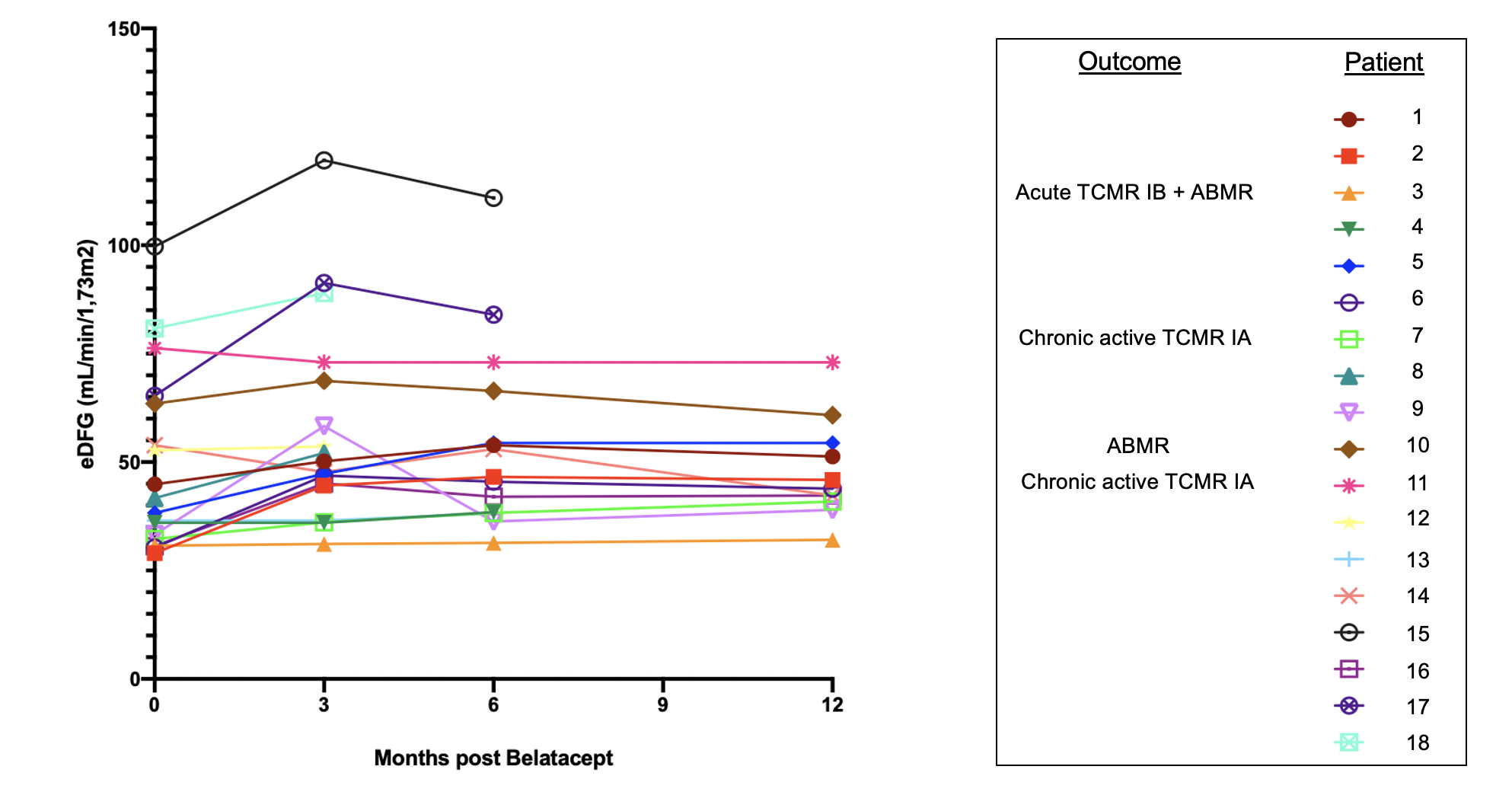
Rejection episodes were observed in 4/18 patients (median 10,3 months, IQR 7,3-19,1): 2 chronic active TCMR IA, 1 mixed acute rejection (TCMR IB and ABMR) and 1 ABMR (both DSA-negative). Among these patients, 2 had prior history of rejection (with normal pre-belatacept biopsies), 1 showed minimal interstitial inflammation without tubulitis (and was off steroids) prior to starting belatacept and 1 had been converted for adherence problems, which subsequently persisted. These rejection episodes showed good evolution after treatment in all patients, but CNI were reintroduced for two of them. Regarding all the other patients, no severe viral complications or development of dnDSA were observed. Four patients had pre-existing DSA which remained stable.
Conclusion: Selected pediatric kidney recipients may benefit from long-term CNI toxicity avoidance, but selection criteria need to be refined to avoid rejection episodes under costimulation blockade.
[1] Vincenti F et al, Belatacept and Long-Term Outcomes in Kidney Transplantation. N Engl J Med 2016
[2] Durrbach A et al, Final results from BENEFIT-EXT, a phase III randomized study. Am J Transplant 2016